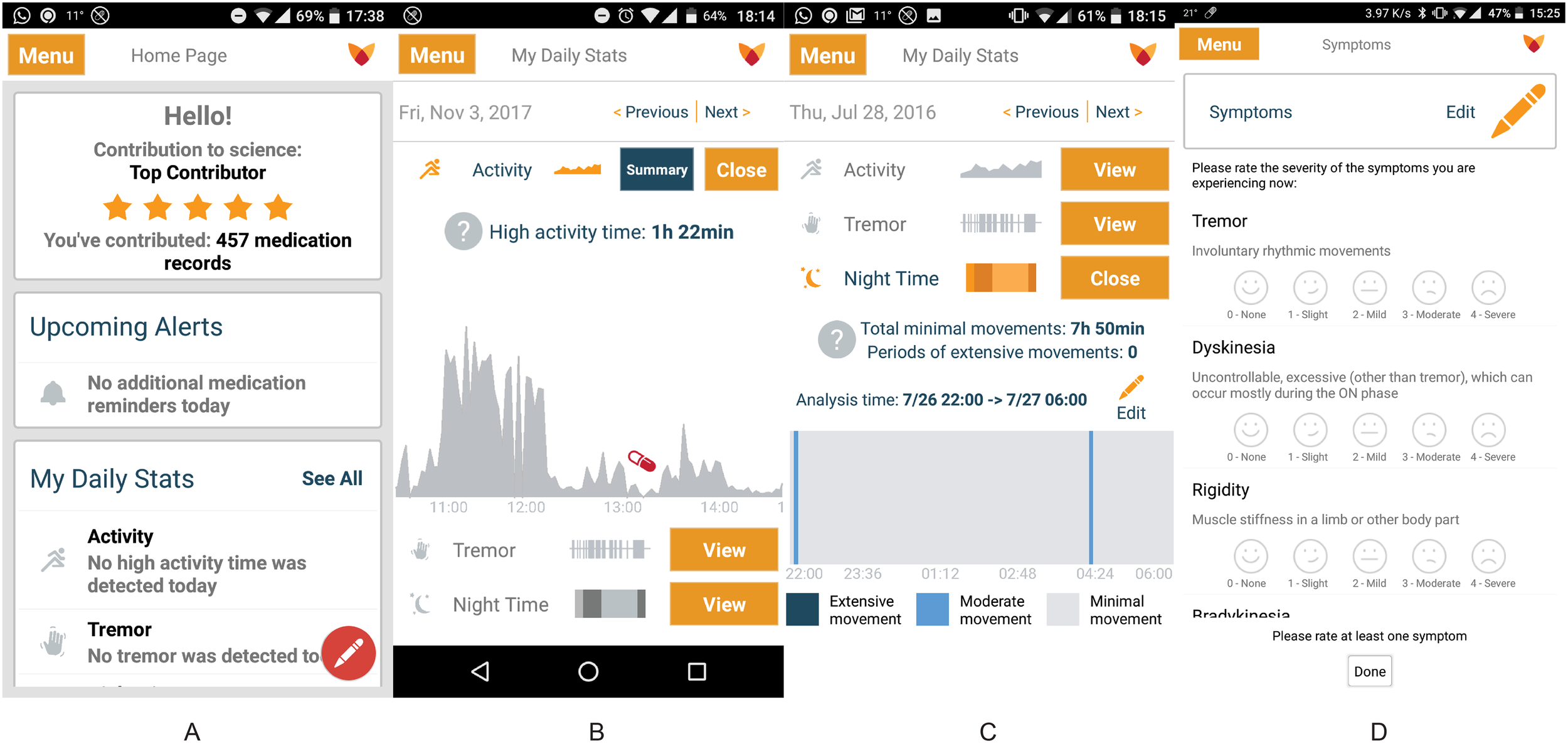
Real World Evidence (RWE) enabled with IoT Wearables
Clinical trials, which seek to prove the safety and efficacy of new treatments through controlled testing on patients, are fraught with challenges that drive up R&D costs and slow the delivery of promising new treatments. The Intel® Pharma Analytics Platform is an edge-to-cloud AI solution that enables continuous capture of clinical data from study subjects using sensors and wearable devices, applying machine learning to objectively measure symptoms and quantify the impact of therapies. So far, the platform has been used in over more than a dozen clinical trials, comprising more than 1.2 million hours of data collection from more than 1,000 patients. Trial results demonstrate increased patient adherence and retention.
Understanding patterns of patient compliance in remote, technology-based clinical trials requires analysis of data collected using digital technologies. The insights from our work suggest that such remote trials are feasible, even when comprising multiple protocol requirements. Beyond our observations, examination of additional factors that impact compliance such as the impact of support, intervention, alternative application design interfaces, and additional covariates can further influence study design.
In the broader perspective, this report supports the growing trend of using mobile applications and wearable technologies to monitor, prompt, and encourage patient compliance with medication intake and performance of clinical assessments. This effort strengthens the notion that these data have the potential to provide further insight regarding patients’ daily lives outside the clinic and potentially evolve into novel endpoints for regulatory purposes.
Read the scientific paper.
Citation: "Characterizing patient compliance over six months in remote digital trials of Parkinson’s and Huntington disease." Shani Cohen, Zeev Waks, Jordan J. Elm, Mark Forrest Gordon, Igor D. Grachev, Leehee Navon-Perry, Shai Fine, Iris Grossman, Spyros Papapetropoulos and Juha-Matti Savola. BMC Medical Informatics and Decision Making. 2018 18:138. https://doi.org/10.1186/s12911-018-0714-7. © The Author(s). 2018
In a recent industry survey for the ICON whitepaper ‘Improving Pharma R&D Efficiency’ respondents cited big data, predictive analytics, smartphones and wearables & sensors as amongst the top disruptive technology trends which will have the greatest impact on clinical trial operations.
So the question remains, in the context of drug development studies, why has the use of this technology been limited to a relatively small number of pilots? It is clear that concerns still remain about implementation of this technology in a clinical trial. These concerns focus on a number of key areas: Patient Acceptance, Device Suitability, Data Complexity and Insight Generation, Operationalisation, Privacy and Security Issues, and Regulatory Acceptance.
Read this article by ICON and Intel Corporation
Authors: Marie McCarthy, Senior Director Product Innovation, ICON; Chen Admati, Head of Intel® Pharma Analytics Platform, Intel
View Transforming Trials Infographic
Read ICON whitepaper “Improving Pharma R&D Efficiency” (registration required)

The Michael J. Fox Foundation and Intel partner to create wearable technology that enables Parkinson's researchers and patients to better understand the disease, slow progression, and improve care to identify a cure.
View the MJFF press release
View the Intel press release
View this brief animation which describes Intel's innovative AI platform for pharmacological clinical trials.
Read the solution brief.
Read this scientific study which assessed the feasibility of using a wearable platform for long-term data collection in a large sample of PD patients, focused on recruitment success, attrition rates, compliance and system usability.
Citation: Silva de Lima AL, Hahn T, Evers LJW, de Vries NM, Cohen E, Afek M, et al. (2017) Feasibility of large-scale deployment of multiple wearable sensors in Parkinson's disease. PLoS ONE 12(12): e0189161. https://doi.org/10.1371/journal.pone.0189161

Parkinson’s disease (PD) — a neurodegenerative disorder that affects movement, cognition, mood and autonomic function — impacts an estimated 5 million people worldwide. Because symptoms vary from individual to individual, research into the disease is further complicated by the lack of objective data. As is typical of many applications used for clinical research, the collection, storage, and analysis of data is complex, expensive, and time-consuming.
The Michael J. Fox Foundation for Parkinson’s Research (MJFF) supported Intel to build a big data analytics platform with AWS infrastructure, using Hadoop and IoT technologies. The platform collects and processes large streams of deidentified sensor data from smartphones and wearable devices, and will make this data available to researchers for analysis. This video highlights how the Foundation is using big data to gain new insights into Parkinson’s disease and further accelerate a cure.
Read the Amazon Web Services blog
Review the Intel AWS Joint IoT Reference Architecture

Intel and the Michael J. Fox Foundation are collaborating to fight against Parkinson ’s disease. In this video, Chen Admati, advanced analytics manager at Intel, explains how consumer health platforms such as wearable technology are helping in research to find a cure.
Read the whitepaper, “Using Wearable Technology to Advance Parkinson’s Research”
Read this scientific study whose objective is to implement and validate quantification of motor dysfunction in a Huntington’s disease (HD) clinical trial using machine learning algorithmic analysis derived from biometric monitoring through a smartphone and wearable sensor combination.
View the Teva Pharmaceuticals press release.
Citation: Implementation and Validation of a Biometric Solution for Remote Monitoring of Motor Symptoms in Patients with Huntington’s Disease in a Phase II Clinical Trial (P2.013). Spyros Papapetropoulos, Shai Fine, Shayna Taylor, Kathleen Blatt, Eli Cohen, Chen Admati, Yonatan Dolan, Josh Lemieux, Igor Grachev, Iris Grossman, Michael Hayden. Neurology Apr 2017, 88 (16 Supplement) P2.013
Learn More
Michael J. Fox Foundation, visit http://michaeljfox.org/
ICON integration of Intel® Pharma Analytics Platform, view press release
Teva Pharmaceuticals, visit www.tevapharm.com
Amazon Web Services and Intel, visit https://aws.amazon.com/intel/
Intel AI in healthcare, view press release
Acknowledgements
Michael J. Fox Foundation: Michael J. Fox, Deborah W. Brooks
Intel: Chen Admati, Yonatan Dolan, Shahar Shpigelman, Itay Yogev, Josh Lemieux, Diane Bryant, Andrew Grove